Before you can apply for an EB-5 visa, you must meet certain requirements. These include the Investment amount, the Legal sources of your investment funds, and the Medical and Immunization examinations. This article will provide you with information on these requirements. Read on to learn how to fulfill them. Then, you can choose the EB 5 regional center where you will invest your funds. If you are still not sure what these requirements are, you can contact consultants.
Investment amount:
When applying for an EB-5 visa, an investor must invest at least US $1.8 million. This investment amount may be lower in certain states, but the total amount must be more than this amount. Additionally, the investment must benefit the domestic and U.S. markets. An immigration lawyer can provide tailored guidance to determine the investment thresholds in your particular case. In addition, they offer comprehensive immigration services.
Legal sources of investment funds:
One of the most important EB-5 visa requirements is the source of funds or legal sources of investment funds. While a loan or gift can be used to meet the investment amount, an EB-5 visa applicant must provide documentation to prove the source of funds. It is possible to use the collateral from your business or other assets to show a legitimate source of funds.
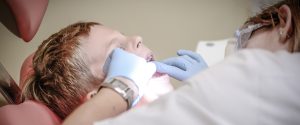
Medical evaluation:
When applying for an EB-5 visa, you must satisfy the Medical evaluation requirements. These exams will determine if you have a communicable disease or any other condition that might render you inadmissible. The medical evaluations must be conducted by a physician designated by the State Department or USCIS. The physician cannot be your family doctor or your primary care physician. If you are not a doctor, you can still submit a medical evaluation to show your qualifications.
Immunizations:
You must have certain vaccinations to be eligible for an EB-5 visa. The Centers for Disease Control and Prevention recently issued Technical Instructions for panel physicians. COVID-19 vaccine is one of the required vaccinations. It is now available for those 16 and older. The vaccine must be approved by the World Health Organization and is approved by the US Food and Drug Administration. A negative result for the vaccine is no longer accepted as proof of immunization.